
09/08/2021
Mechanism that triggers brain neurone response revealed
It is the first time it has been possible to see how neurotransmitters and proteins interact at the atomic level to trigger neuronal responses.
Researchers from the G-protein-coupled receptor-based drug discovery research group of GRIB (IMIM-UPF), led by Dr. Jana Selent have been able to verify, with a degree of precision never before achieved, how the process that triggers the response of neurones in the brain occurs. This is an essential mechanism for understanding how moods or even processes such as addictions are produced, and in which neurotransmitters, molecules that help transmit information between neurones through specialised receptors, the G protein-coupled receptors (GPCRs), play a vital role.
"Neurotransmission is one of the most crucial physiological processes, as its dysregulation can result in various neuropsychiatric disorders", explains Dr. Jana Selent, principal author of the study, published in the journal Chemical Science, and coordinator of the research group that led the work. Very small changes in how information is transmitted by these molecules can trigger different reactions in the brain, some of which are linked to behaviour, addictions and moods.
Reference article: Stepniewski TM, Mancini A, Agren R, Torrens-Fontanals M, M Semache, Bouvier M, Sahlholm K, Breton B, Selent J. Mechanistic insights into dopaminergic and serotonergic neurotransmission-concerted interactions with helices 5 and 6 drive the functional outcome. Chem Sci, 2021 DOI: 10.1039/D1SC00749A.
_1625659045.jpg)
7/7/2021
RISK-HUNT3R project launches: risk assessment of chemicals integrating human centric next generation testing strategies promoting the 3Rs
RISK-HUNT3R is the new European effort hosting world-leading experts from many different disciplines aiming to develop a novel and modular framework for animal-free next generation risk assessment (NGRA). The Integrative Bioinformatics and PharmacoInformatics groups of GRIB, led by Ferran Sanz and Manuel Pastor respectively, take part of this project. Also the company MedBioinformatics Solutions, an spin-off of the GRIB, is partner of RISKHUNT3R.
Over the past decades, science and society have increasingly started demanding a paradigm shift towards chemical safety and risk assessment without using animals. In addition to growing ethical concerns around the use of animal studies, there is the fundamental need to address all potential health effects relevant and specific to humans. Furthermore, the limited efficiency of animal-based testing (low throughput, high costs, insufficient laboratory capacity) will not be able to keep up the pace in ensuring the required protection of European citizens based on the sheer number of chemicals that require safety assessment. The vision of the RISK-HUNT3R consortium is to establish a reliable and cost-effective chemical risk assessment strategy based entirely on non-animal approaches.

06/07/2021
Thesis defence of Joaquim Aguirre: "In silico tools to study diseases and polypharmacology through the lens of network medicine"
Next Wednesday 14th of July at 11:00, Joaquim Aguirre, member of the Structural Bioinformatics group of GRIB will read his thesis "In silico tools to study diseases and polypharmacology through the lens of network medicine
The event will be online. Free access at https://us02web.zoom.us/j/83124036086

22/06/2021
GRIB participates in a new world-wide initiative that paves the way for the large-scale annotation and characterization of small proteins in the genome
The first results of the international 'Non-canonical ORF Working Group' to annotate novel translated open reading frames (ORFs) in the human genome have been made publicly available. The initiative includes researchers from 37 different Institutions, including Mar Albà, ICREA researcher at the Research Programme on Biomedical Informatics (GRIB, IMIM-UPF), and former PhD student, Jorge Ruiz-Orera. The latter researcher, now at the Max Delbrück Center, co-leads the project together with researchers at the European Bioinformatics Institute and the Broad Institute. The effort also has the support of Gencode/Ensembl, HGNC and Uniprot.
The researchers have collected data from already published studies and built a consensus list that contains 7,264 new translated ORFs, including many ORFs in long non-coding RNAs, as well as upstream ORFs (uORFs) in coding transcripts. The list will be accessible from the Gencode web page and, in subsequent phases of the project, it will be part of standard gene annotations. The project paves the way for the large-scale annotation and characterization of small proteins that have remained hidden in the genome. A more complete annotation of the human proteome will be key to identify mutations that inactivate small proteins and cause disease.
Preprint: Mudge, Ruiz-Orera, Prensner et al. A community-driven roadmap to advance research on translated open reading frames detected by Ribo-seq. bioRxiv June 10, 2021.
28/06/2021
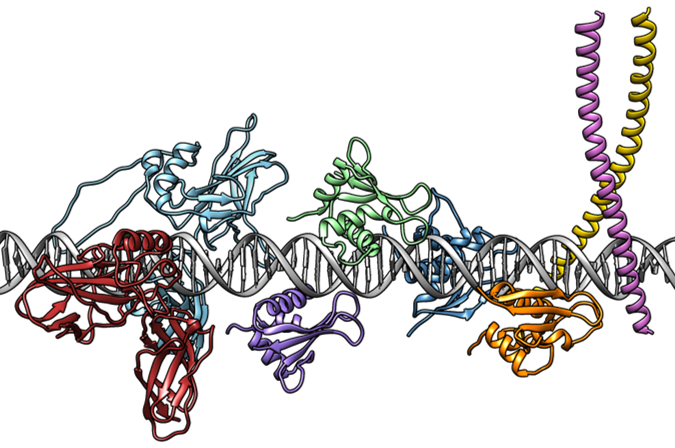
Thesis defence of Alberto Meseguer: "Methods to model and assess protein-DNA and protein-protein interactions in the context of gene regulation".
Next Friday, 2nd of July at 10:00, Alberto Meseguer, member of the Structural Bioinformatics group of GRIB will read his thesis "Methods to model and assess protein-DNA and protein-protein interactions in the context of gene regulation".
The event will be online. Free access at:https://us02web.zoom.us/j/8198872626

22/06/2021
Open GRIB seminar on June, 22nd: Bioinformatics, combining facility and research by Lara Nonell
The last Open GRIB seminar took place on 22/06/2021 at 10:00 h with the title "Bioinformatics, combining facility and research", by Dr Lara Nonell, member of MARGenomics at IMIM and researcher of the Human Computational Biology group of the Programme on Biomedical Informatics-GRIB (IMIM/UPF).
Abstract
Bioinformatics is a common need in most research labs. Therefore it is important to have access to a core facility that is able to implement the standard methodology to extract knowledge from biological data, including omics data. In addition, new methods are continuously developed in the field to meet increasing data analysis demands. In this context, we will explain our experience as a service oriented facility and will also see some of the recently funded projects we are involved in, coming out of fields as disparate as COVID-19, bladder cancer or major depression disorder.
26/05/2021
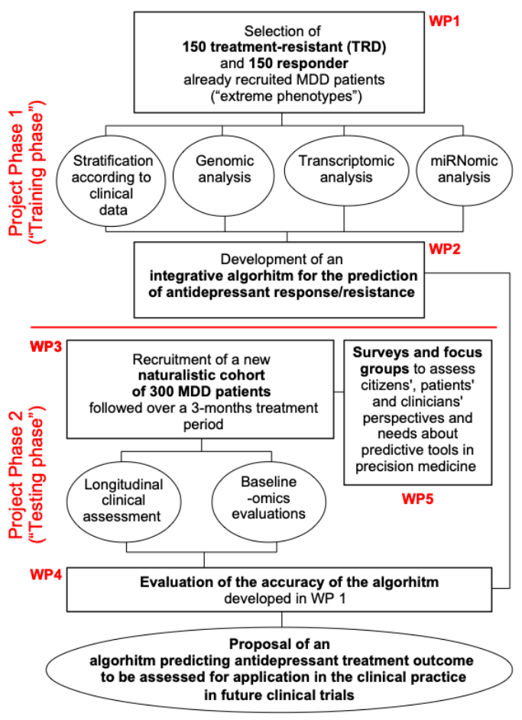
Proyecto PROMPT: Hacia una medicina de precisión para la predicción de la respuesta al tratamiento del trastorno depresivo mayor
El GRIB inicia este nuevo proyecto liderado por la Universidad de Muenster (Alemania) en el que participan los investigadores Ferran Sanz, Lara Nonell y Laura I. Furlong. El objetivo es desarrollar herramientas predictivas que faciliten la toma de decisiones en el tratamiento del trastorno depresivo mayor, mejorando la prevención y gestión de esta enfermedad.
El trastorno depresivo mayor (TDM) es la enfermedad psiquiátrica más común en todo el mundo con un enorme impacto socioeconómico. La farmacoterapia representa la primera opción de tratamiento; sin embargo, esta solo tiene éxito en alrededor del 33% de los pacientes mientras un 30% se manifiestan resistentes al tratamiento lo cual se asocia con características clínicas o genéticas específicas.
Este proyecto representa una prueba de concepto. Los resultados obtenidos proporcionarán información sobre la viabilidad y utilidad del enfoque propuesto, con el objeto de diseñar ensayos clínicos futuros en los que los algoritmos puedan ser probados como una herramienta predictiva para apoyar la toma de decisiones de los médicos, lo que permitirá una mejor prevención y gestión de la resistencia al TDM.

20/04/2021
Open GRIB Seminars: join the first one on April, 27th at 10:00: "Taking full advantage of Real World Data through Biomedical Informatics"
We are going to start a series of seminars to show the research that the groups of GRIB are doing. Every two months one group will present their research to the other groups so that we can learn what is being done next door. Of course the seminars will also be open to other researchers outside the Programme.
The first seminar will take place next Tuesday, April 27th at 10:00 AM. The speaker, Miguel Angel Mayer, Senior Researcher of the Integrative Biomedical Informatics Group of GRIB will talk about 'Taking full advantage of Real World Data through Biomedical Informatics'.
The seminar will take 1 hour approximately, you can join it at this link https://www.gotomeet.me/GRIB/opengrib_seminars
We wish to see you all there!
13/04/2021
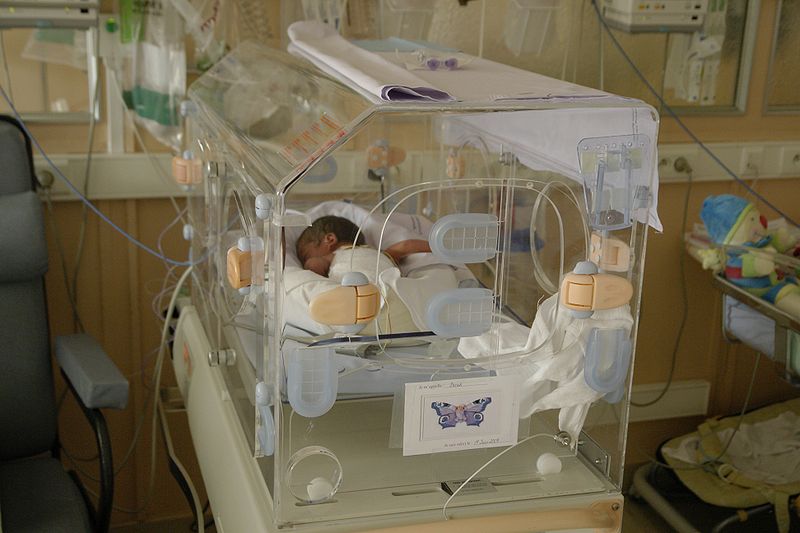
Postnatal molecular changes associated with the fetal inflammatory response have been identified in extremely preterm newborns
The molecular changes observed after birth reveal for the first time a postnatal alteration of adaptive immunity in extremely preterm newborns affected by fetal inflammatory response before birth.
A new study provides the largest catalogue to date of postnatal molecular changes associated with the fetal inflammatory response in extremely preterm newborns. The study, published in The FEBS Journal, has been jointly coordinated by Robert Castelo, associate professor at UPF and head of the Functional Genomics group of GRIB, and Teresa Cobo, a specialist in maternal-fetal medicine at Hospital Clínic-IDIBAPS, with Daniel Costa, a paediatrician at the Fundació Salut Empordà as a first author.
"This research has allowed us to identify postnatal molecular changes associated with the fetal inflammatory response that provide clues on the mechanisms that trigger systemic inflammation", explains Robert Castelo. "A step forward in this research is to look up earlier whether we can detect these molecular changes, during pregnancy, in the amniotic fluid of women at greater risk of developing an intraamniotic infection. This research opens a window of opportunity for these vulnerable newborns as it enables redirecting their monitoring to have a positive influence on the neonatal prognosis", says Teresa Cobo. "The investigation was the result of an intense, selfless collaboration among professionals in a variety of fields, and it has been made possible thanks to the generosity of the families involved. The results of the study may help the long road to improving the health of preterm newborns and their families", Daniel Costa concludes.
Article Reference: Costa D, Bonet N, Solé A, González de Aledo-Castillo JM, Sabidó E, Casals F, Rovira C, Nadal A, Marin JL, Cobo T, Castelo R. Genome-wide postnatal changes in immunity following fetal inflammatory response. FEBS J, 2021; 288(7): 2311-2331. doi: 10.1111/febs.15578.

15/02/2021
The effects of treatment with antidepressant drugs evaluated through the analysis of patients’ tweets
Scientists from the Integrative Biomedical Informatics research group have identified behavioural and linguistic changes in tweets in Spanish published by users suffering from depression and who are taking medication to treat this disease.
Their work has been published in Journal of Medical Internet Research and was led by Ferran Sanz; with Angela Leis and Francesco Ronzano as first authors, who conducted the work together with Miguel Angel Mayer and Laura I Furlong, all from the Integrative Biomedical Informatics research group.
Depression is one of the most widespread mental illnesses. According to the World Health Organization, it affects more than 322 million people of all ages and is one of the leading causes of disability worldwide.
Reference article: Leis A, Ronzano F, Mayer MA, Furlong LI, Sanz F. Evaluating Behavioral and Linguistic Changes During Drug Treatment for Depression Using Tweets in Spanish: Pairwise Comparison Study. J Med Internet Res. 2020 Dec. doi: 10.2196/20920.



